how to read an rf factor in chromatography In thin-layer chromatography, the retention factor (Rf) is used to compare and help identify compounds. The Rf value of a compound is equal to the distance traveled by the compound . What do the Animal Crossing NFC Cards do? The Animal Crossing NFC cards .
0 · why rf value not good
1 · why is rf importnat chemistry
2 · why are rf values useful
3 · what is rf value chromatography
4 · what does rf value mean
5 · rf value in paper chromatography
6 · how to measure rf values
7 · how to calculate rf values
Ali Express is the cheapest you will find them. Gen4 is the best and it's gonna cost you about .
It is possible to determine the characteristic rate of movement of each substance on the chromatography paper as the moving phase moves at a certain temperature and for a specific solvent. This is represented by the R f value, which stands for relative front or retardation factor.
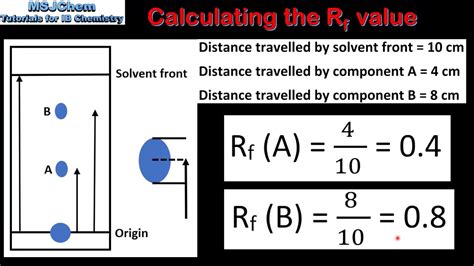
A convenient way for chemists to report the results of a TLC plate in lab notebooks is through a "retention factor",\(^2\) or \(R_f\) value, which quantitates a compound's movement (Equation \ref{2}).
In this article we will learn about Rf value also known as Retention Factor. Here we will discuss calculation of Rf values and its importance in thin layer chromatography. Key .
Learn about the Rf value, a key parameter in chromatography that allows for the identification and analysis of individual components in a mixture. Discover why the Rf value is important, how it's .
In thin-layer chromatography, the retention factor (Rf) is used to compare and help identify compounds. The Rf value of a compound is equal to the distance traveled by the compound .
Retention Factor. After a separation is complete, individual compounds appear as spots separated vertically. Each spot has a retention factor (Rf) which is equal to the distance migrated over the total distance covered by .In planar chromatography in particular, the retardation factor R F is defined as the ratio of the distance traveled by the center of a spot to the distance traveled by the solvent front. [2] .It is possible to determine the characteristic rate of movement of each substance on the chromatography paper as the moving phase moves at a certain temperature and for a specific solvent. This is represented by the R f value, which stands for relative front or retardation factor.A convenient way for chemists to report the results of a TLC plate in lab notebooks is through a "retention factor",\(^2\) or \(R_f\) value, which quantitates a compound's movement (Equation \ref{2}).
In this article we will learn about Rf value also known as Retention Factor. Here we will discuss calculation of Rf values and its importance in thin layer chromatography. Key words: Rf value or Retention factor, Thin layer chromatography (TLC), . In thin layer chromatography, retention factor (Rf) is the distance that a compound travels through the stationary phase (TLC plate) between the origin spot and the distance the solvent front moved above the origin.Learn about the Rf value, a key parameter in chromatography that allows for the identification and analysis of individual components in a mixture. Discover why the Rf value is important, how it's calculated, and the factors that influence it.
In thin-layer chromatography, the retention factor (Rf) is used to compare and help identify compounds. The Rf value of a compound is equal to the distance traveled by the compound divided by the distance traveled by the solvent front (both measured from the origin). Retention Factor. After a separation is complete, individual compounds appear as spots separated vertically. Each spot has a retention factor (Rf) which is equal to the distance migrated over the total distance covered by the solvent. The \( R_f\) formula is \[ R_f= \dfrac{\text{distance traveled by sample}}{\text{distance traveled by solvent}} \]In planar chromatography in particular, the retardation factor R F is defined as the ratio of the distance traveled by the center of a spot to the distance traveled by the solvent front. [2] Ideally, the values for R F are equivalent to the R values used in column chromatography. [2]Rf values, or the Retention Factor, is a ratio used to describe the relationship between the distance moved by components in a mixture relative to the distance moved by the solvent. It is calculated by dividing the distance moved by the component .
Learn how to calculate and interpret RF values in chromatography with our comprehensive educational guide. Master the principles of RF values and their significance in analytical chemistry.
smart card jobs in singapore
It is possible to determine the characteristic rate of movement of each substance on the chromatography paper as the moving phase moves at a certain temperature and for a specific solvent. This is represented by the R f value, which stands for relative front or retardation factor.
A convenient way for chemists to report the results of a TLC plate in lab notebooks is through a "retention factor",\(^2\) or \(R_f\) value, which quantitates a compound's movement (Equation \ref{2}). In this article we will learn about Rf value also known as Retention Factor. Here we will discuss calculation of Rf values and its importance in thin layer chromatography. Key words: Rf value or Retention factor, Thin layer chromatography (TLC), .
why rf value not good
In thin layer chromatography, retention factor (Rf) is the distance that a compound travels through the stationary phase (TLC plate) between the origin spot and the distance the solvent front moved above the origin.Learn about the Rf value, a key parameter in chromatography that allows for the identification and analysis of individual components in a mixture. Discover why the Rf value is important, how it's calculated, and the factors that influence it.In thin-layer chromatography, the retention factor (Rf) is used to compare and help identify compounds. The Rf value of a compound is equal to the distance traveled by the compound divided by the distance traveled by the solvent front (both measured from the origin).
Retention Factor. After a separation is complete, individual compounds appear as spots separated vertically. Each spot has a retention factor (Rf) which is equal to the distance migrated over the total distance covered by the solvent. The \( R_f\) formula is \[ R_f= \dfrac{\text{distance traveled by sample}}{\text{distance traveled by solvent}} \]In planar chromatography in particular, the retardation factor R F is defined as the ratio of the distance traveled by the center of a spot to the distance traveled by the solvent front. [2] Ideally, the values for R F are equivalent to the R values used in column chromatography. [2]Rf values, or the Retention Factor, is a ratio used to describe the relationship between the distance moved by components in a mixture relative to the distance moved by the solvent. It is calculated by dividing the distance moved by the component .
why is rf importnat chemistry
why are rf values useful
Posted on Nov 1, 2021 12:10 PM. On your iPhone, open the Shortcuts app. Tap on the Automation tab at the bottom of your screen. Tap on Create Personal Automation. Scroll down and select NFC. Tap on Scan. Put .
how to read an rf factor in chromatography|how to calculate rf values